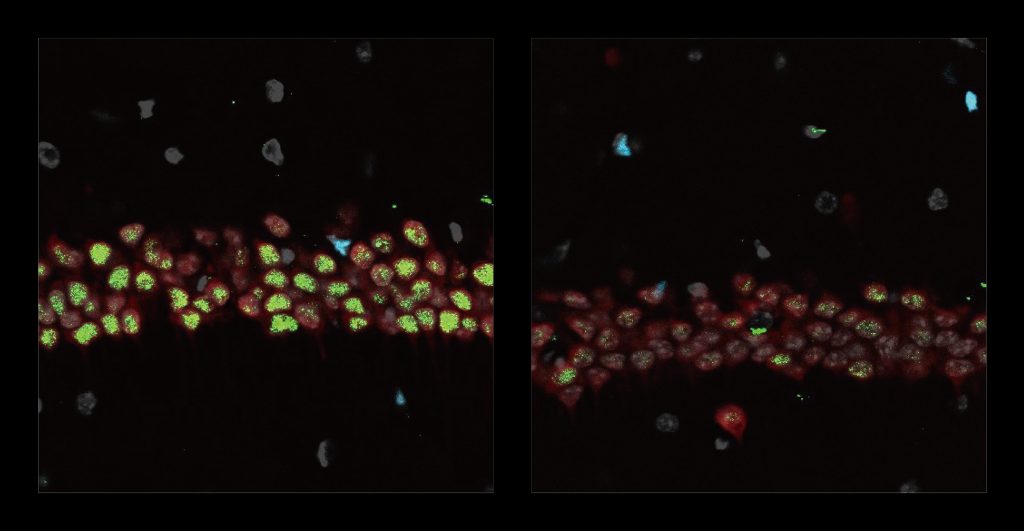
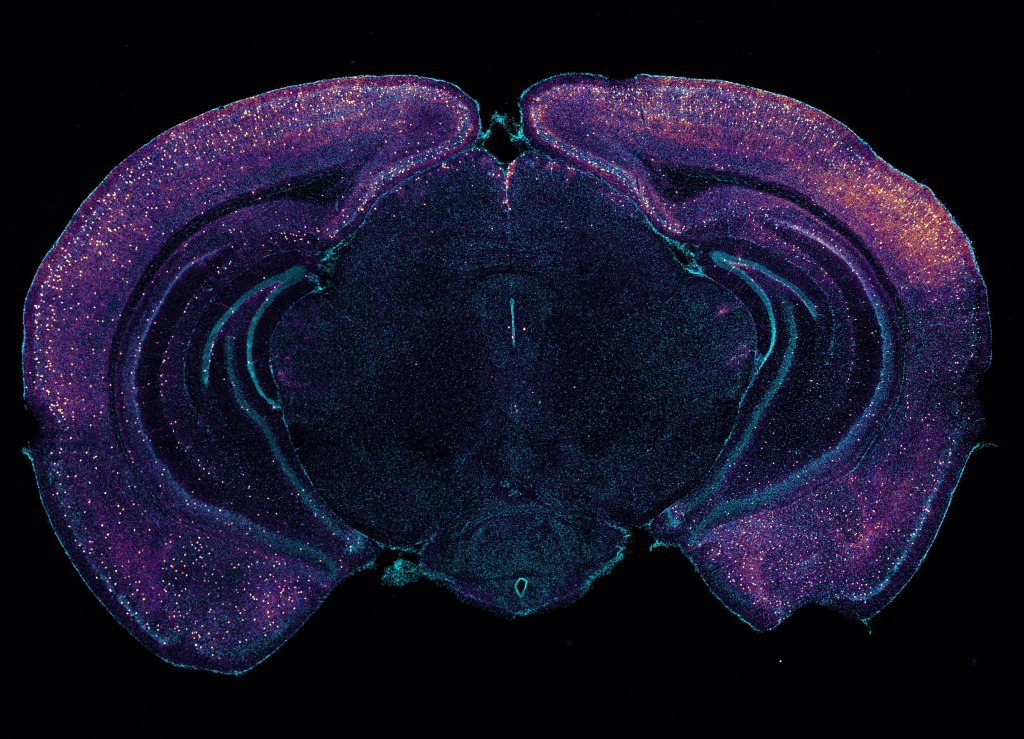
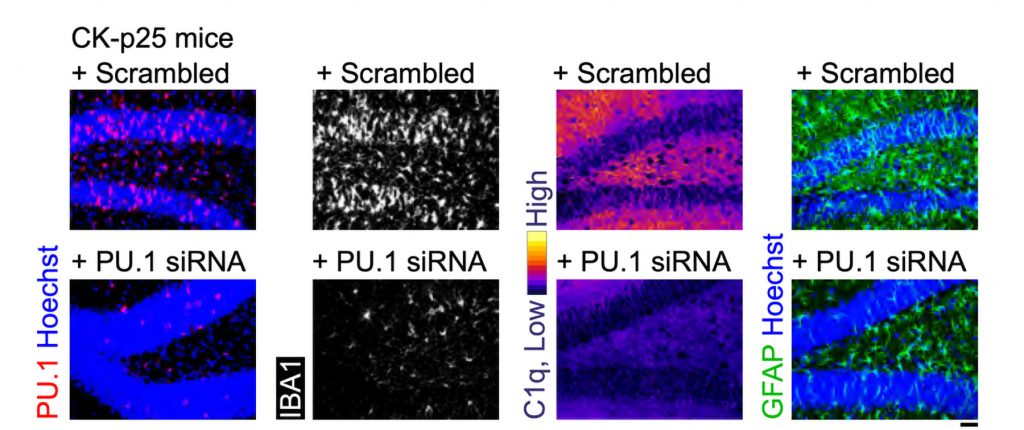
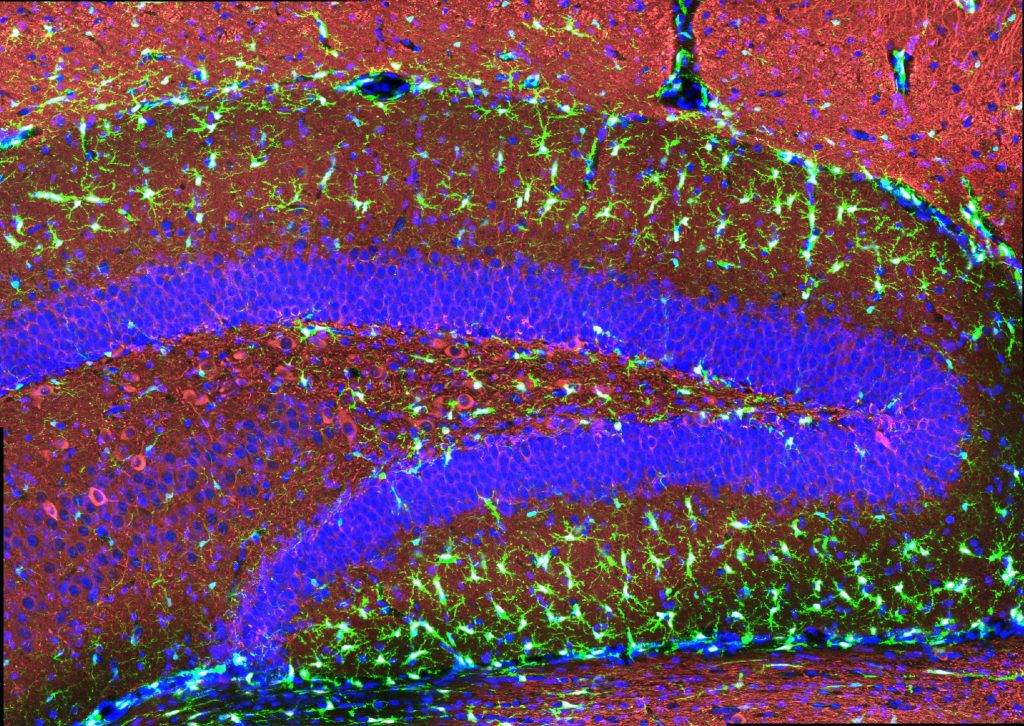
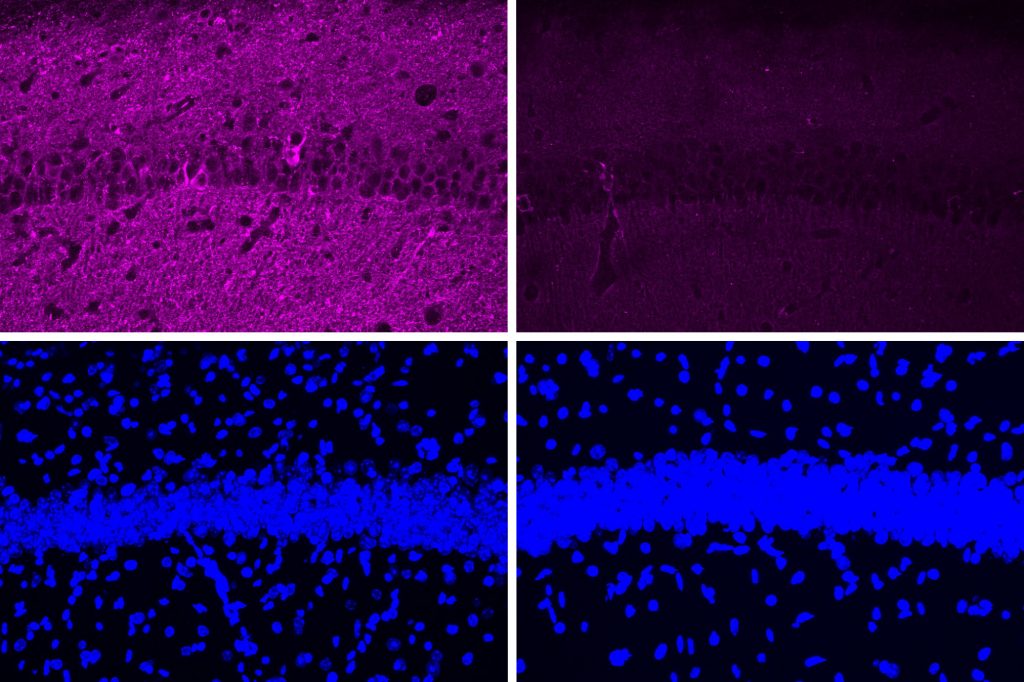
Stimulating gamma brain waves may protect cancer patients from memory impairment and other cognitive effects of chemotherapy. Patients undergoing chemotherapy often experience cognitive effects such as memory impairment and difficulty concentrating — a condition commonly known as “chemo brain.” MIT researchers have now shown that a noninvasive treatment that stimulates gamma frequency brain waves may …